|
This is required by the law of conservation of matter as well. For the following molecules or ions (where the central atom is underlined): i. A pair of dots between chemical symbols for. 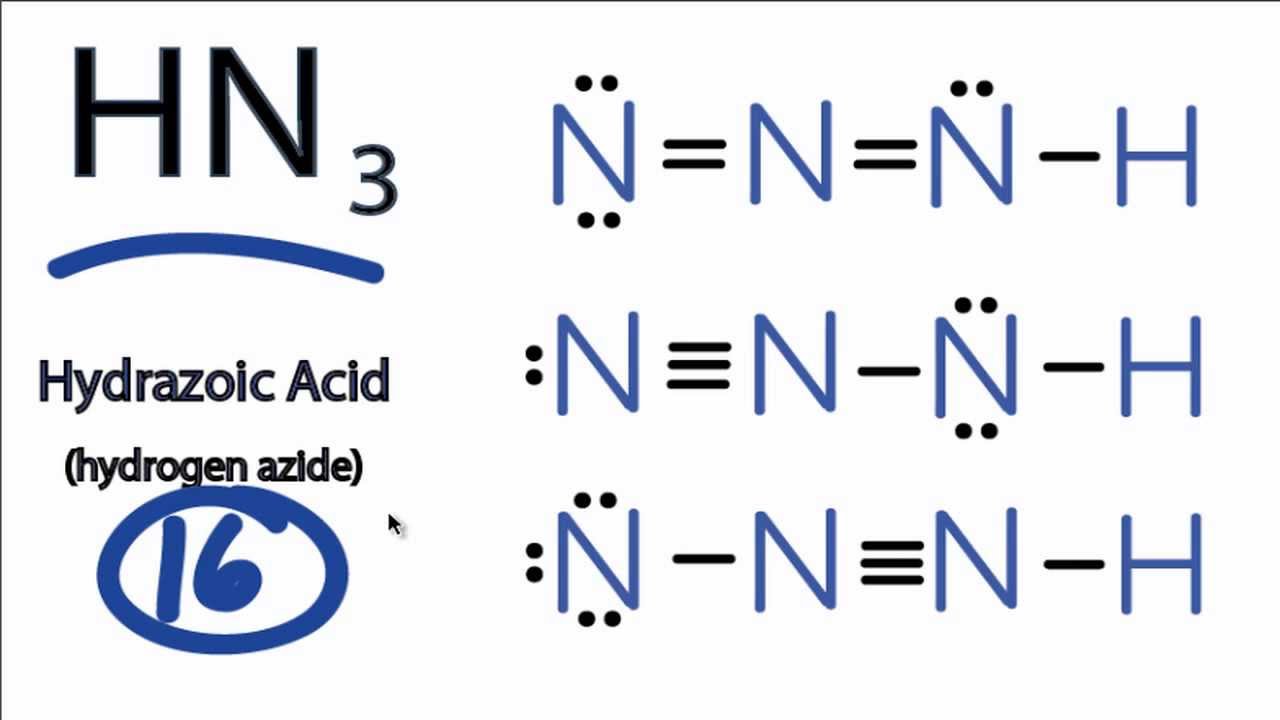
For example, when two chlorine atoms, each with 7 valence electrons, come together to form a diatomic chlorine molecule, the Lewis structure shows that there will be a sharing of two electrons between the two. In Lewis dot structures each dot represents an electron. Using Lewis dot structures and the octet rule, we can predict and represent the electronic structure of covalently bonded molecules. Lewis structures are a useful way to summarize certain information about bonding and may be thought of as electron bookkeeping. The need for the number of electrons lost being equal to the number of electrons gained explains why ionic compounds have the ratio of cations to anions that they do. Lewis dot structures reflect the electronic structures of the elements, including how the electrons are paired. 
These three ions attract each other to give an overall neutral-charged ionic compound, which we write as Na 2O. What we need is a second Na atom to donate a second electron to the O atom: The O atom still does not have an octet of electrons.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
